$330.00
GLP-3 RT is a lab-made tri-agonist peptide that works on three hormone receptors: GLP-1, GIP, and glucagon. This triple effect makes it a useful tool for researchers studying metabolism, energy use, and weight control. Scientists use GLP-3 RT to see how the body manages blood sugar, fat use, and heart function in lab tests.
In stock
GLP-3 RT is a lab-made tri-agonist peptide that works on three hormone receptors: GLP-1, GIP, and glucagon. This triple effect makes it a useful tool for researchers studying metabolism, energy use, and weight control. Scientists use GLP-3 RT to see how the body manages blood sugar, fat use, and heart function in lab tests.
Preclinical and clinical studies, like LY3437943 and TRIUMPH-3, show how GLP-3RT works on several hormone receptors at the same time. This lets researchers watch how tri-agonist peptides affect hormone signals, energy balance, and heart health. These studies give new ideas for treating obesity and related health issues.
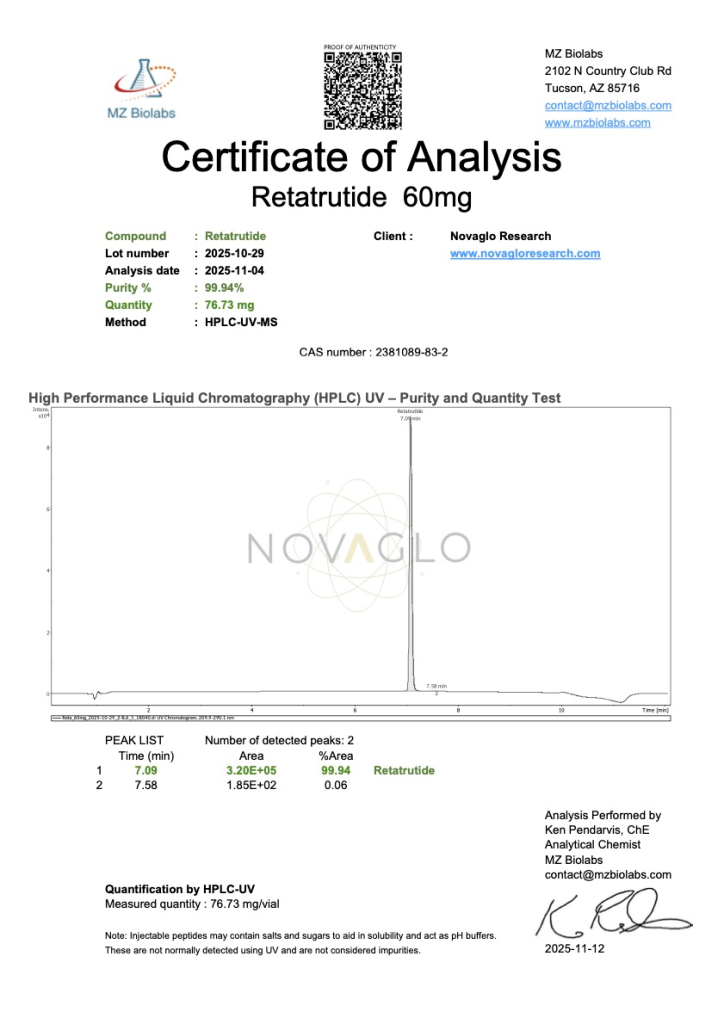
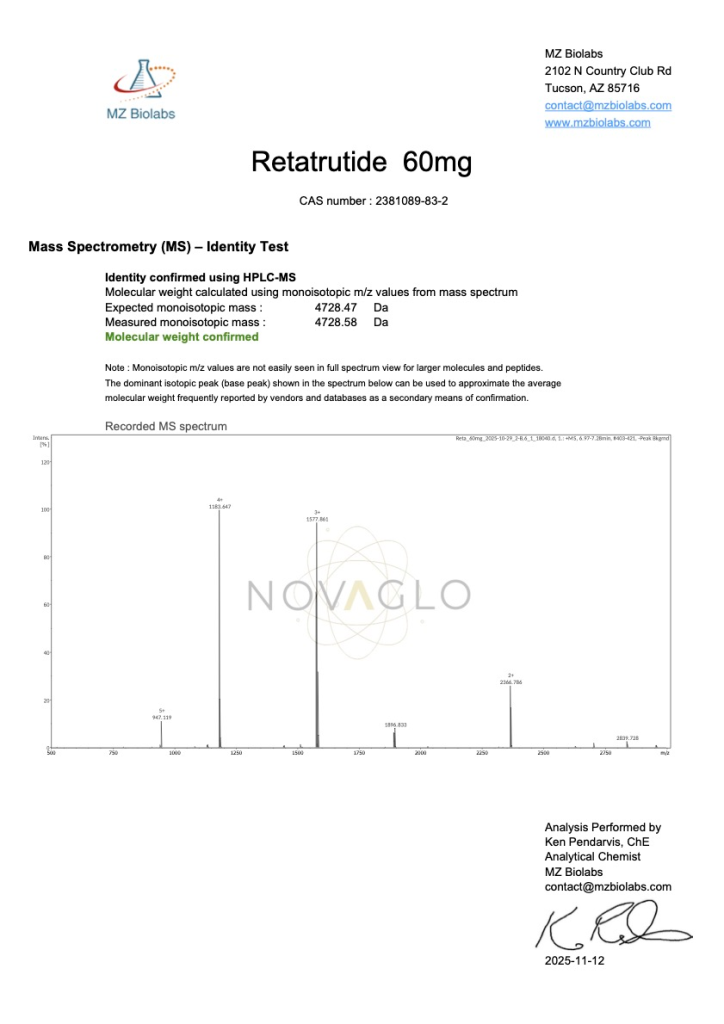
NOVAGLO Peptides are manufactured in trusted cGMP-certified facilities located in the USA and worldwide, delivering >99% purity with third-party Certificates of Analysis (COA) completed on every lot.
Peptide Research Team—Your Exclusive 30% Black Friday Savings with code PRT30—when you spend $300 or more